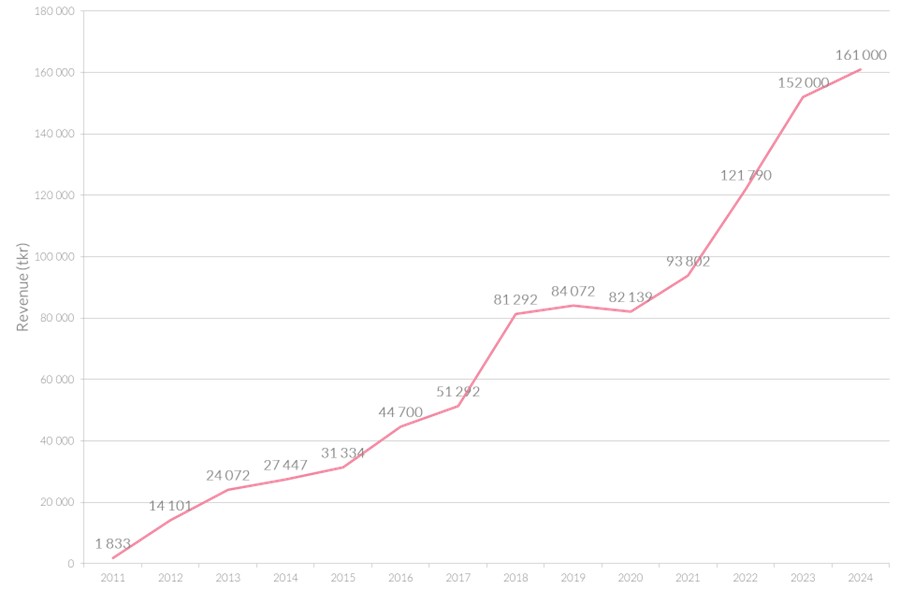
Scandinavian Biopharma Reports All-Time High Revenue for 2024
Scandinavian Biopharma is pleased to announce another year of strong growth, strategic milestones and key achievements. For the full year 2024, the company’s revenue reached 161 MSEK, reflecting continued momentum in both its distribution and development business.
The company’s distribution business achieved its strongest sales performance to date, driven by the introduction of new products and an expanded geografical presence. A major highlight in 2024 was the signing of a joint procurement agreement with the European Commission to supply diphtheria antitoxin, reinforcing Scandinavian Biopharma’s role as a reliable supplier of specialty biopharma products to the European market.
In vaccine development, the ETEC vaccine candidate successfully completed its Pediatric Phase IIb trial in The Gambia, involving 4,936 children aged 6–18 months. The study evaluated safety, immunogenicity and protective efficacy against moderate to severe diarrhoea caused by ETEC, and preliminary results are highly encouraging. Preparations for the upcoming Phase III studies are progressing as planned. A pre-study for a planned Phase III CHIM (Controlled Human Infection Model) study in the USA was initiated to verify the attack rate of the proposed challenge dose.
The company is on track to register the world’s first vaccine against ETEC-related diarrhoea, targeting both children and adults in endemic regions, as well as travellers to high-risk destinations.
With a record year behind it, Scandinavian Biopharma is entering 2025 with strong momentum and a clear focus on advancing its vaccine programs and specialty biopharma offerings.